We all know about batteries. We also accustom ourselves to how they work. Batteries make sure our portable electronics (phones, laptops, etc.) are in full charge. But, most of us don’t understand how a battery works. It would help if you didn’t worry, though. This article simplifies and explains everything you need to know about battery separators.
Let’s dive in.
Contents
- 1 What Exactly Is A Battery Separator?
- 2 What Is the Function of a Battery Separator?
- 3 What’s the Structure of a Battery Separator?
- 4 Which Materials Make Battery Commercial Separators?
- 5
- 6 Manufacturing Process for a Battery Separator
- 7 What Are the Properties of a Battery Separator?
- 8 Challenges (and Solutions) with Battery Separators?
- 9 Are There Any Developments in Lithium Ion Battery?
- 10 Conclusion
What Exactly Is A Battery Separator?
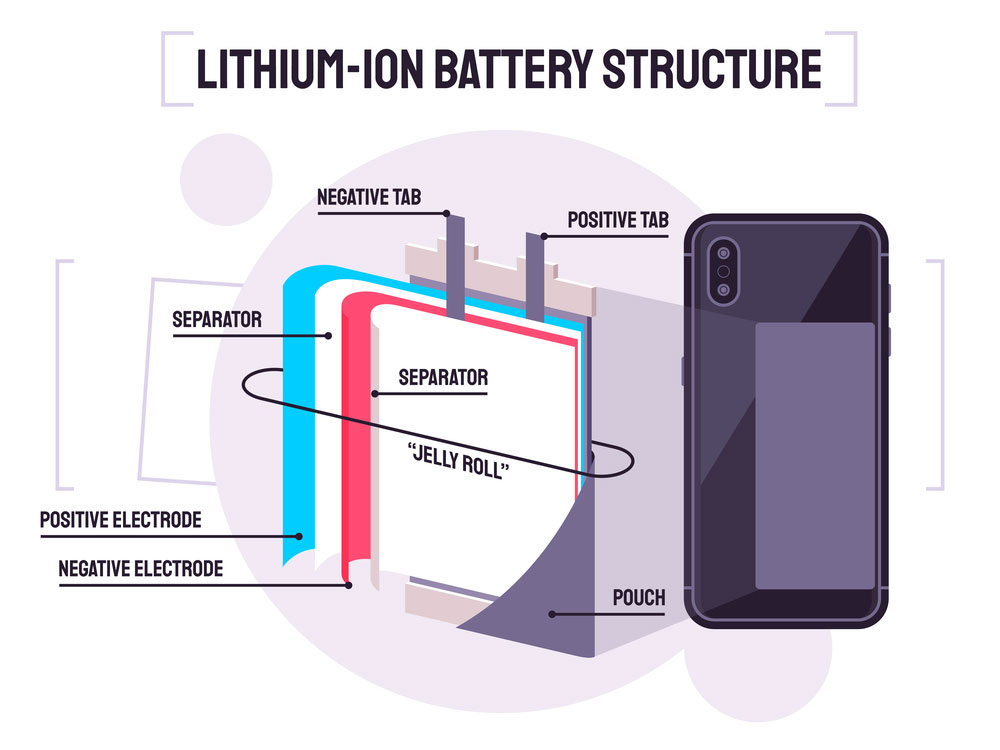
Lithium-ion Battery Structure
A battery separator is a permeable membrane between its anode and cathode. The two are the battery’s electrodes.
The separator keeps both electrodes apart to avoid an electrical short circuit. The anode is the positive electrode, and the cathode is the negative electrode.
Also, the separator is a critical component. It acts as a passage of electrons to and from either the cathode or the anode. The battery separator must be porous to allow transportation of the lithium ions.
The performance and efficiency of Lithium-ion batteries rely on separator properties and structure.
What Is the Function of a Battery Separator?
A battery separator’s function is to guarantee safety by avoiding short-circuit.
But that’s not all.
The main role of a battery separator is to allow the safe movement of ions. This is what makes the battery charge up your electronic device.
The movement of electrons from the anode to the cathode happens when the battery is charging. And when the electrons move in the reverse, from cathode to anode, the battery is discharging.
What’s the Structure of a Battery Separator?
The structure is all about three sections – the cathode, the anode, and a porous separator. Simple, right?
Let’s look at the picture below.
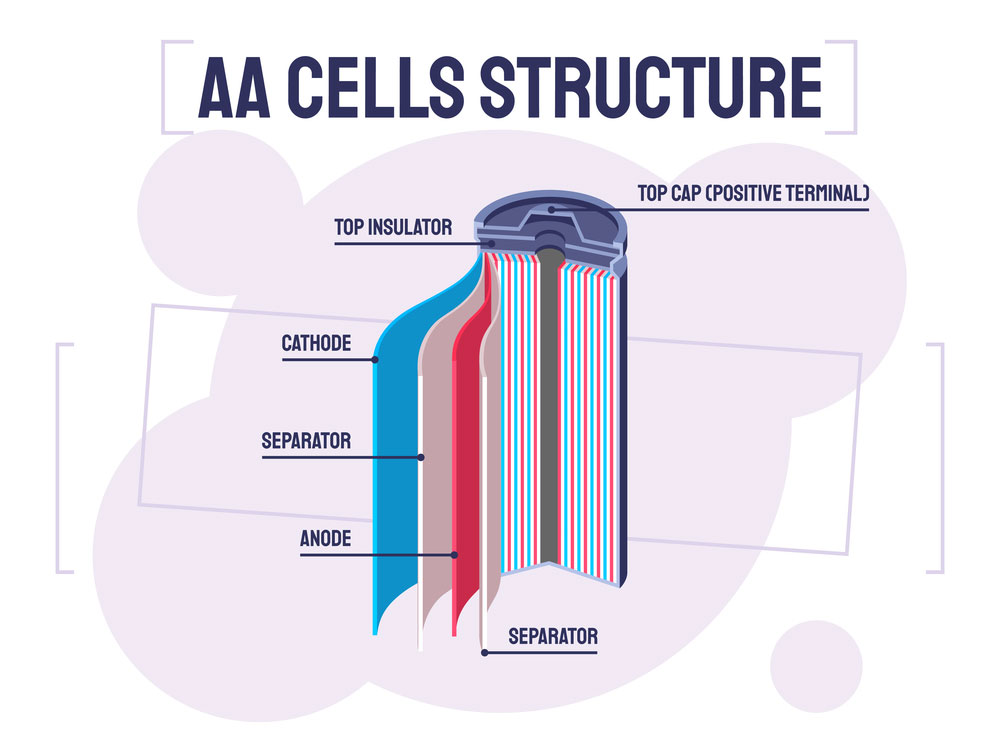
AA Cells Structure
The electron movement is from either anode or cathode electrodes. The cathode is the negative electrode, while the anode is the positive electrode, depending on the battery’s charging or discharging roles.
The separator is porous to allow electron movement. For efficiency, the separator usually consists of polymeric membranes that form micro-porous layers.
Although the pores are not visible to the human eye, they allow the movement of lithium ions.
Without the separator, a battery’s functionality is void. The porous polyolefin films control the leakage of ions when an auxiliary battery is ideal (self-discharge).
The microporous layer doesn’t allow electrical conductivity, thereby always acting as an isolator.
The only note is that temperature affects the separators’ performance. If the temperature rises to a certain point, the pores block and shut down. Hence, it stops the movement of lithium ions.
Which Materials Make Battery Commercial Separators?
The material needs to be a non-conductor. And should have great thermal stability (explained later in this article).
Manufacturers use special polyolefin grades to produce rechargeable lithium-ion batteries. The polyolefin material comes about by laminating polyethylene and polypropylene together.
Polyolefin is a preference because of its mechanical properties, chemical stability. Companies also prefer polyolefin due to its low cost.
The image below is a polyolefin battery separator, thus applicable in rechargeable batteries.

Source: Plasticstoday.com
Apart from polyolefin, other materials include:
- Polyvinyl chloride
- Nylon ceramic coating layer
- Polyester
- Asbestos
- Glass and
- Tetrafluoroethylene.
Manufacturing Process for a Battery Separator
Like its structure, the process of producing battery separators is simple. The process is by stretching or wet processing the polyolefin material.
The dry procedure involves using a mechanical force to create the pores. And is suitable for higher power densities.
The wet process involves adding additives to the polyolefin film material. Thus, it is a chemical process of creating pores.
While simple, the dry process causes the pores to have different sizes. The result is a reduction in the separator’s effectiveness. The dry procedure also weakens the separator, thus minimizing its puncture strength.
The wet process is a little bit more complicated – and expensive too. Luckily, it creates the same pore sizes for the separator. The outcome is improvement in wettability.
Side note – Wettability is the ability of the battery separator to “wet” with its electrolyte solutions. While the wet process is effective, it doesn’t increase electrolyte retention.
What Are the Properties of a Battery Separator?
The question helps understand what makes a good auxiliary battery separator. The separator is not only responsible for the battery’s efficiency but also for safety.
We best understand both requirements (efficiency and safety) by examining their different properties. So let’s have a go.
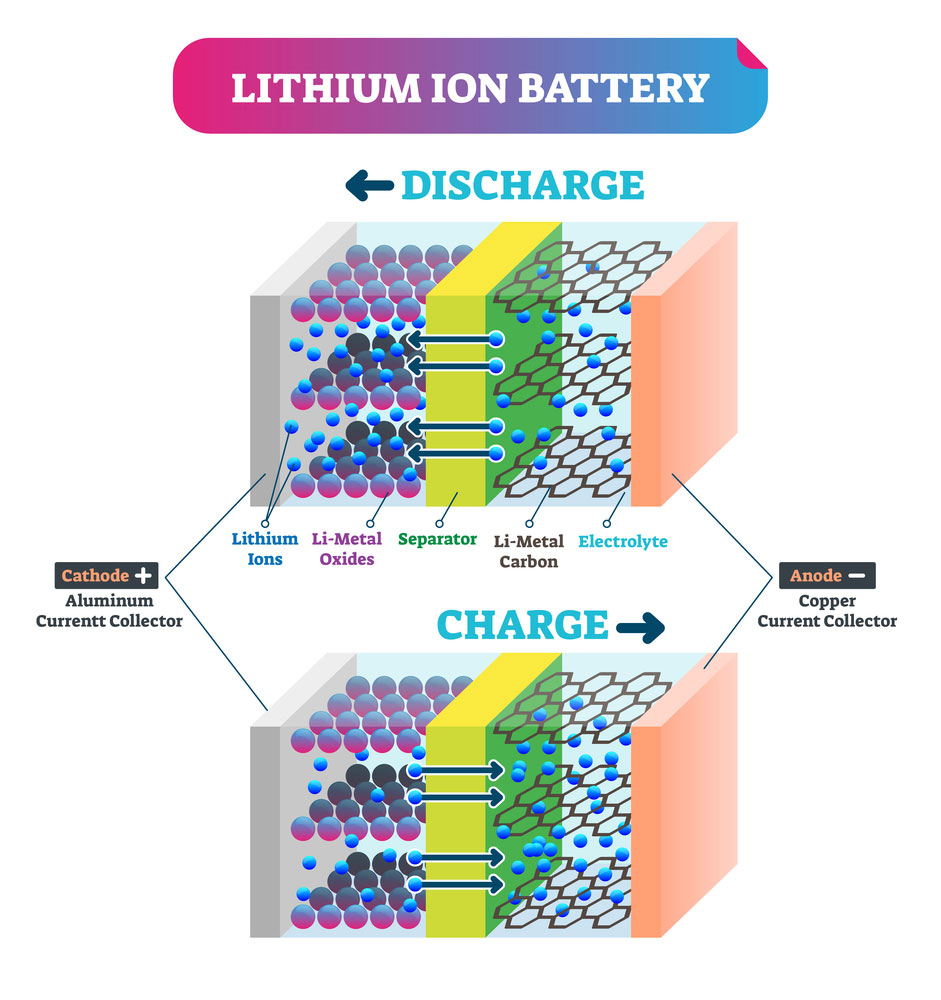
Lithium-Ion battery
1. Chemical Stability
As you already know, a battery separator ought to have non-conductivity properties. That is, the separator shouldn’t react with either anode or cathode electrodes.
Also, the separator needs to stay chemically stable. Once again, this makes sure the separator doesn’t react with the liquid electrolyte. Such stability helps the battery overcome degradation.
2. Thickness and Mechanical Strength
The goal is to develop a thin battery without losing its mechanical strength. In other words, when a Li-Ion battery separator is thin, it helps ease its power and energy density.
With this in mind, battery manufacturers ensure the primary battery has sufficient tensile strength. It helps prevent the battery’s stretching, especially during a winding process.
3. Porosity and Pore Density
The intention is for the separator to have a high pore density. This holds electrolytes and allows free movement of lithium ions between the electrodes.
The separator’s porosity shouldn’t be too large, not too small. The pore size should be wide enough to close whenever the battery shuts down.
We measure a separator’s porosity in percentage. The average porosity is 40 percent. And yes, the pores should have a uniform distribution.
4. Thermal Stability
The separator needs to endure and withstand a wide range of temperatures. It shouldn’t curl or pucker in very high temperatures. The separator should shut down at temperatures lower than the polymer’s melting point before the thermal runaway. In essence, this is the annealing process.
Challenges (and Solutions) with Battery Separators?
Okay, there are constant developments to improve the performance of rechargeable Li-ion batteries. But, that’s met with quite a several challenges.
Most of these challenges are due to emerging needs. For instance, there are needs and requirements for batteries to withstand high temperatures.
Also, there is a need for batteries to last long without compromising their cell properties.
Some of the challenges facing the manufacture of battery separators include:
a) The need to produce thinner separators
b) To increase wettability
c) The intention to improve the battery’s performance in high temperatures
Let me explain these challenges further.
The need for stable thermal performance is due to the invention of power grids and electric cars. Both inventions require heavy-duty batteries that can withstand high temperatures.
Manufacturers are improving cell performance by using new materials. The thermal stabilities are better than polyolefin.
The need for enhanced wettability is due to the polyolefin’s incompatibility. The choice of polymer is not compatible with common electrolyte materials.
The new, conventional electrolytes are characteristic of high dielectric constants. Which helps shorten the battery’s manufacturing process.
At the same time, polyolefin’s incompatibility results in the uneven current distribution. The result is a primary battery that doesn’t charge your electric device long enough.
Finally, there is always a demand to have thinner batteries. As expected, this is challenging. Batteries need thicker electrodes to hold more charge. Nonetheless, companies are coming up with special polyolefin grades to improve Lid’s high molecular weight.
Are There Any Developments in Lithium Ion Battery?
Manufacturers burn the midnight oil to improve the performance of battery separators.
Companies develop unique (and possibly patented) ways to enhance wettability and thermal stability. Let’s have a look at some of the improvements so far.

Lithium-Ion battery
a) Decorating the Separator with Silica Nanoparticles
The process involves attaching silica nanoparticles to the separators’ pores and pore walls. The expecting result is an improved wettability towards the battery’s electrolyte. The method also enhances the battery’s resistance to heat.
While the separator material heightens heat resistance, the silica nanoparticles boost wettability.
b) Using Phase Inversion Process to Produce Novel PEIs (Polyetherimides)
The process involves using PD and BPADA to produce separators. It is helpful because the battery separator now has better properties. For instance, the separator can handle a wide range of conductivities.
The PEIs also help minimize the battery’s degree of swelling. Thus, it results in fast electrolyte wicking.
Better yet, the battery separator displays thermal stability of up to 220°C.
c) Hydrogen Induced Crosslinking (HHC)
The technique is a covalent crosslinking of a polyethylene oxide onto a PP separator. The effect is an increase in the electrolyte affinity of the polyolefin.
With such a modification, Li-ion batteries have high capacity retention. The batteries also have low internal resistance.
Conclusion
As we’ve seen, a battery separator is a permeable membrane that acts as an isolator. It separates the cathode (negative electrode) from the anode (positive electrode).
The separator is a vital material because it determines the battery’s effectiveness. With a lowly reliable separator, a secondary battery is an equally low quality.
Battery manufacturers are discovering new battery separator technology to enhance its performance. For instance, such secondary batteries are applicable in PCBs (printed circuit boards), available at WellPCB.





